Preparation for radioiodine therapy in patients with differentiated thyroid cancer: a modern perspective (a review)
- Authors: Reinberg M.V.1, Slashchuk K.Y.1, Trukhin A.A.1, Avramova K.I.1, Sheremeta M.S.1
-
Affiliations:
- Endocrinology Research Centre
- Issue: Vol 4, No 4 (2023)
- Pages: 543-568
- Section: Reviews
- URL: https://ogarev-online.ru/DD/article/view/262966
- DOI: https://doi.org/10.17816/DD532728
- ID: 262966
Cite item
Abstract
Thyroid cancer is the most prevalent tumor of the endocrine system, accounting for 1%–3% of all malignant neoplasms as of 2021. Differentiated forms, papillary and follicular, with a relatively favorable prognosis, are detected in 90% of cases. The combination of surgical treatment, subsequent suppressive hormonal therapy, and radioiodine therapy provides a favorable prognosis in patients with differentiated thyroid cancer. However, an insufficient response to radioiodine therapy may be possible, which may be associated with multiple factors, including the preparation step for radioiodine therapy. To date, the question of choosing the optimal method of patient preparation remains relevant. This paper presents a review of the scientific literature on the preparation of patients with differentiated thyroid cancer for radioiodine therapy. The principles of preparation are based on the recommendations of leading expert societies, and publications related to this topic are highlighted and summarized, including the adverse events associated with radioiodine therapy, quality of life, efficacy, and long-term results of treatment. The main purpose of this review was to provide a comprehensive insight into the methods of preparing a patient with differentiated thyroid cancer for radioiodine therapy, highlight existing problems and promising areas of research, and modernize treatment toward personalized therapy. Scientific articles and reviews from the National Library of Medicine, Cochrane Library, and Google Scholar databases, published up to the end of January 2023, were searched by the keywords listed below in their various combinations. Recommendations from the following scientific communities were used: Russian Clinical Guidelines for Differentiated Thyroid Cancer, American Thyroid Association, European Thyroid Association, National Comprehensive Cancer Network, European Association of Nuclear Medicine, British Thyroid Association, and European Society for Medical Oncology. Articles not available in full, not in English or Russian, or systematic reviews of a similar topic, were excluded. In total, 124 sources were selected and analyzed, general tendencies of modern approaches to preparation for radioiodine therapy and actual problems were highlighted, concepts of optimization of preparation for radioiodine therapy within the framework of personalized therapy were covered, and results and conclusions were presented.
Full Text
##article.viewOnOriginalSite##About the authors
Maria V. Reinberg
Endocrinology Research Centre
Author for correspondence.
Email: mrezerford12@gmail.com
ORCID iD: 0009-0002-1632-2197
ResearcherId: IUO-4237-2023
Russian Federation, Moscow
Konstantin Y. Slashchuk
Endocrinology Research Centre
Email: slashuk911@gmail.com
ORCID iD: 0000-0002-3220-2438
SPIN-code: 3079-8033
MD, Cand. Sci. (Med.)
Russian Federation, MoscowAlexey A. Trukhin
Endocrinology Research Centre
Email: Alexey.trukhin12@gmail.com
ORCID iD: 0000-0001-5592-4727
SPIN-code: 4398-9536
Cand. Sci. (Engin.)
Russian Federation, MoscowKarina I. Avramova
Endocrinology Research Centre
Email: dravramovak@gmail.com
ORCID iD: 0009-0008-4970-8911
SPIN-code: 4330-0263
Russian Federation, Moscow
Marina S. Sheremeta
Endocrinology Research Centre
Email: marina888@yandex.ru
ORCID iD: 0000-0003-3785-0335
SPIN-code: 7845-2194
MD, Cand. Sci. (Med.)
Russian Federation, MoscowReferences
- Vanushko VE, Tsurkan AYu. Treatment of differentiated thyroid cancer: cureunt statement of the problem. Clinical and experimental thyroidology. 2010;6(2):24–33. (In Russ). doi: 10.14341/ket20106224-33
- Kaprin AD, Starinskii VV, Shakhzadova AO, editors. Zlokachestvennye novoobrazovaniya v Rossii v 2021 godu (zabolevaemost’ i smertnost’). Moscow: MNIOI im. P.A. Gertsena −of NMRRC of the Ministry of Health of Russia; 2022. (In Russ).
- Durante C, Haddy N, Baudin E, et al. Long-term outcome of 444 patients with distant metastases from papillary and follicular thyroid carcinoma: benefits and limits of radioiodine therapy. The Journal of Clinical Endocrinology & Metabolism. 2006;91(8):2892–2899. doi: 10.1210/jc.2005-2838
- Cancer Stat Facts: Thyroid Cancer [Internet]. National Cancer Institute: Surveillance, Epidemiology, and End Results Program. [cited 2023 Sep 1]. Available from: https://seer.cancer.gov/statfacts/html/thyro.html
- Braverman LE, Cooper DS, Kopp P. Werner & Ingbar’s The Thyroid: A Fundamental and Clinical Text. 11th ed.. Philadelphia, PA: Wolters Kluwer; 2021.
- Hassan A, Razi M, Riaz S, et al. Survival Analysis of Papillary Thyroid Carcinoma in Relation to Stage and Recurrence Risk: A 20-Year Experience in Pakistan. Clinical Nuclear Medicine. 2016;41(8):606–613. doi: 10.1097/RLU.0000000000001237
- Well-differentiated thyroid cancer. Clinical guidelines. ID 329. Approved by the Scientific and Practical Council of the Ministry of Health of the Russian Federation. 2020. Available from: https://cr.minzdrav.gov.ru/recomend/329_1 (In Russ)
- Avram AM, Giovanella L, Greenspan B, et al. SNMMI Procedure Standard/EANM Practice Guideline for Nuclear Medicine Evaluation and Therapy of Differentiated Thyroid Cancer: Abbreviated Version. Journal of Nuclear Medicine. 2022;63(6):15N–35N.
- Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26(1):1–133. doi: 10.1089/thy.2015.0020
- Pacini F, Fuhrer D, Elisei R, et al. 2022 ETA Consensus Statement: What are the indications for post-surgical radioiodine therapy in differentiated thyroid cancer? European Thyroid Journal. 2022;11(1). doi: 10.1530/etj-21-0046
- Filetti S, Durante C, Hartl D, et al. Thyroid cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology. 2019;30(12):1856–1883. doi: 10.1093/annonc/mdz400
- Perros P, Boelaert K, Colley S, et al. Guidelines for the management of thyroid cancer. Clinical Endocrinology. 2014;81 Suppl. 1:1–122. doi: 10.1111/cen.12515
- Haddad RI, Bischoff L, Ball D, et al. Thyroid Carcinoma, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. Journal of the National Comprehensive Cancer Network. 2022;20(8):925–951. doi: 10.6004/jnccn.2022.0040
- Golger A, Fridman TR, Eski S, et al. Three-week thyroxine withdrawal thyroglobulin stimulation screening test to detect low-risk residual/recurrent well-differentiated thyroid carcinoma. Journal of Endocrinological Investigation. 2003;26(10):1023–1031. doi: 10.1007/bf03348202
- Davids T, Witterick IJ, Eski S, et al. Three-Week Thyroxine Withdrawal: A Thyroid-Specific Quality of Life Study. The Laryngoscope. 2006;116(2):250–253. doi: 10.1097/01.mlg.0000192172.61889.43
- Lee J, Yun MJ, Nam KH, et al. Quality of life and effectiveness comparisons of thyroxine withdrawal, triiodothyronine withdrawal, and recombinant thyroid-stimulating hormone administration for low-dose radioiodine remnant ablation of differentiated thyroid carcinoma. Thyroid. 2010;20:173–179. doi: 10.1089/thy.2009.0187
- Leboeuf R, Perron P, Carpentier AC, Verreault J, Langlois MF. L-T3 preparation for whole-body scintigraphy: a randomized-controlled trial. Clinical Endocrinology. 2007;67(6):839–844. doi: 10.1111/j.1365-2265.2007.02972.x
- Rajamanickam S, Chaukar D, Siddiq S, Basu S, D’Cruz A. Quality of life comparison in thyroxine hormone withdrawal versus triiodothyronine supplementation prior to radioiodine ablation in differentiated thyroid carcinoma: a prospective cohort study in the Indian population. European Archives of Oto-Rhino-Laryngology. 2021;279(4). doi: 10.1007/s00405-021-06948-6
- Luna R, Penín M, Seoane I, et al. ¿Es necesario suspender durante 4 semanas el tratamiento con tiroxina antes de la realización de un rastreo-ablación? Endocrinología y Nutrición. 2012;59(4):227–231. (In Spanish). doi: 10.1016/j.endonu.2012.02.004
- Dow KH, Ferrell BR, Anello C. Quality-of-life changes in patients with thyroid cancer after withdrawal of thyroid hormone therapy. Thyroid. 1997;7(4):613–619. doi: 10.1089/thy.1997.7.613
- Liel Y. Preparation for radioactive iodine administration in differentiated thyroid cancer patients. Clinical Endocrinology. 2002;57(4):523–527. doi: 10.1046/j.1365-2265.2002.01631.x
- Piccardo A, Puntoni M, Ferrarazzo G, et al. Could short thyroid hormone withdrawal be an effective strategy for radioiodine remnant ablation in differentiated thyroid cancer patients? European Journal of Nuclear Medicine and Molecular Imaging. 2018;45(7):1218–1223. doi: 10.1007/s00259-018-3955-x
- Santos PA, Flamini ME, Mourato FA, et al. Is a four-week hormone suspension necessary for thyroid remnant ablation in low and intermediate risk patients? A pilot study with quality-of-life assessment. Brazilian Journal of Radiation Sciences. 2022;10(4):1–16. doi: 10.15392/2319-0612.2022.2047
- Cooper DS, Doherty GM, Haugen BR, et al. Revised American thyroid association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19(11):1167–1214. doi: 10.1089/thy.2009.0110
- Rosário PW, Vasconcelos FP, Cardoso LD, et al. Managing thyroid cancer without thyroxine withdrawal. Arquivos Brasileiros de Endocrinologia & Metabologia. 2006;50(1):91–96. doi: 10.1590/s0004-27302006000100013
- Edmonds CJ, Hayes S, Kermode JC, Thompson BD. Measurement of serum TSH and thyroid hormones in the management of treatment of thyroid carcinoma with radioiodine. The British Journal of Radiology. 1977;50(599):799–807. doi: 10.1259/0007-1285-50-599-799
- Giovanella L, Piccardo A. A “new/old method” for TSH stimulation: could a third way to prepare DTC patients for 131I remnant ablation possibly exist? European Journal of Nuclear Medicine and Molecular Imaging. 2015;43(2):221–223. doi: 10.1007/s00259-015-3245-9
- Semenov DYu, Boriskova ME, Farafonova UV, et al. Prognostic value of Sodium-Iodide Symporter (NIS) in differentiated thyroid cancer. Clinical and experimental thyroidology. 2015;11(1):50. (In Russ). doi: 10.14341/ket2015150-58
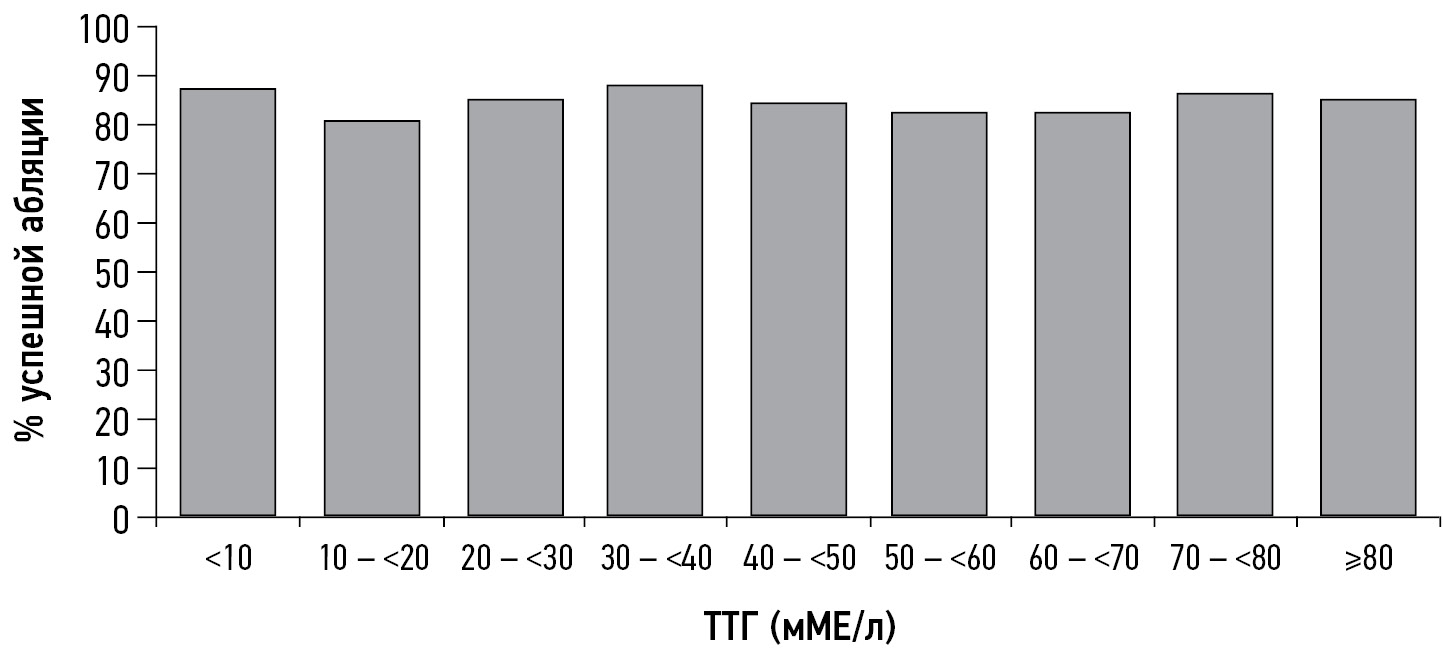
- Xiao J, Yun C, Cao J, et al. A pre-ablative thyroid-stimulating hormone with 30-70 mIU/L achieves better response to initial radioiodine remnant ablation in differentiated thyroid carcinoma patients. Scientific Reports. 2021;11(1). doi: 10.1038/s41598-020-80015-8
- Zhao T, Liang J, Guo Z, Li T, Lin Y. In Patients with Low- to Intermediate-Risk Thyroid Cancer, a Preablative Thyrotropin Level of 30 μIU/mL Is Not Adequate to Achieve Better Response to 131I Therapy. Clinical Nuclear Medicine. 2016;41(6):454–458. doi: 10.1097/rlu.0000000000001167
- Hasbek Z, Turgut B. Is Very High Thyroid Stimulating Hormone Level Required in Differentiated Thyroid Cancer for Ablation Success? Molecular Imaging and Radionuclide Therapy. 2016;25(2):79–84. doi: 10.4274/mirt.88598
- Vrachimis A, Riemann B, Mäder U, Reiners C, Verburg FA. Endogenous TSH levels at the time of 131I ablation do not influence ablation success, recurrence-free survival or differentiated thyroid cancer-related mortality. European Journal of Nuclear Medicine and Molecular Imaging. 2016;43(2):224–231. doi: 10.1007/s00259-015-3223-2
- Montesano T, Durante C, Attard M, et al. Age influences TSH serum levels after withdrawal of l-thyroxine or rhTSH stimulation in patients affected by differentiated thyroid cancer. Biomedicine & Pharmacotherapy. 2007;61(8):468–471. doi: 10.1016/j.biopha.2007.04.001
- Ju N, Hou L, Song H, et al. TSH ≥30 mU/L may not be necessary for successful 131I remnant ablation in patients with differentiated thyroid cancer. European Thyroid Journal. 2023;12(4). doi: 10.1530/ETJ-22-0219
- Ren B, Zhu Y. A New Perspective on Thyroid Hormones: Crosstalk with Reproductive Hormones in Females. International Journal of Molecular Sciences. 2022;23(5):2708. doi: 10.3390/ijms23052708
- Rubio GA, Catanuto P, Glassberg MK, Lew JI, Elliot SJ. Estrogen receptor subtype expression and regulation is altered in papillary thyroid cancer after menopause. Surgery. 2018;163(1):143–149. doi: 10.1016/j.surg.2017.04.031
- Derwahl M, Nicula D. Estrogen and its role in thyroid cancer. Endocrine-Related Cancer. 2014;21(5):T273–T283. doi: 10.1530/erc-14-0053
- Rajoria S, Suriano R, Shanmugam A, et al. Metastatic phenotype is regulated by estrogen in thyroid cells. Thyroid. 2010;20(1):33–41. doi: 10.1089/thy.2009.0296
- Tala H, Robbins R, Fagin JA, Larson SM, Tuttle RM. Five-year survival is similar thyroid cancer patients with metastases prepared for radioactive iodine therapy with either thyroid hormone withdrawal or recombinant human TSH. The Journal of Clinical Endocrinology & Metabolism. 2011;96(7):2105–2111. doi: 10.1210/jc.2011-0305
- Rosario PW, Mourão GF, Calsolari MR. Recombinant human TSH versus thyroid hormone withdrawal in adjuvant therapy with radioactive iodine of patients with papillary thyroid carcinoma and clinically apparent lymph node metastases not limited to the central compartment (cN1b). Archives of Endocrinology and Metabolism. 2017;61(2):167–172. doi: 10.1590/2359-3997000000247
- Hugo J, Robenshtok E, Grewal R, Larson S, Tuttle RM. Recombinant human thyroid stimulating hormone-assisted radioactive iodine remnant ablation in thyroid cancer patients at intermediate to high risk of recurrence. Thyroid. 2012;22(10):1007–1015. doi: 10.1089/thy.2012.0183
- Robenshtok E, Tuttle RM. Role of Recombinant Human Thyrotropin (rhTSH) in the Treatment of Well-Differentiated Thyroid Cancer. Indian Journal of Surgical Oncology. 2012;3(3):182–189. doi: 10.1007/s13193-011-0115-1
- Higuchi CRS, Fernanda P, Jurnior PA, et al. Clinical Outcomes After Radioiodine Therapy, According to the Method of Preparation by Recombinant TSH vs. Endogenous Hypothyroidism, in Thyroid Cancer Patients at Intermediate-High Risk of Recurrence. Frontiers in Nuclear Medicine. 2021;1. doi: 10.3389/fnume.2021.785768
- Lawhn-Heath C, Flavell RR, Chuang EY, Liu C. Failure of iodine uptake in microscopic pulmonary metastases after recombinant human thyroid-stimulating hormone stimulation. World Journal of Nuclear Medicine. 2020;19(1):61–64. doi: 10.4103/wjnm.WJNM_29_19
- Lee H, Paeng JC, Choi H, et al. Effect of TSH stimulation protocols on adequacy of low-iodine diet for radioiodine administration. PLoS One. 2021;16(9):e0256727. doi: 10.1371/journal.pone.0256727
- Driedger AA, Kotowycz N. Two Cases of Thyroid Carcinoma That Were Not Stimulated by Recombinant Human Thyrotropin. The Journal of Clinical Endocrinology & Metabolism. 2004;89(2):585–590. doi: 10.1210/jc.2003-031650
- Mernagh P, Campbell S, Dietlein M, et al. Cost-effectiveness of using recombinant human TSH prior to radioiodine ablation for thyroid cancer, compared with treating patients in a hypothyroid state: The German perspective. European Journal of Endocrinology. 2006;155(3):405–414. doi: 10.1530/eje.1.02223
- Mernagh P, Suebwongpat A, Silverberg J, Weston A. Cost-effectiveness of using recombinant human thyroid-stimulating hormone before radioiodine ablation for thyroid cancer: The Canadian perspective. Value in Health. 2010;13(3):180–187. doi: 10.1111/j.1524-4733.2009.00650.x
- Borget I, Bonastre J, Catargi B, et al. Quality of life and cost-effectiveness assessment of radioiodine ablation strategies in patients with thyroid cancer: results from the randomized phase III ESTIMABL trial. Journal of Clinical Oncology. 2015;33(26):2885–2892. doi: 10.1200/JCO.2015.61.6722
- Nijhuis TF, van Weperen W, de Klerk JMH. Costs associated with the withdrawal of thyroid hormone suppression therapy during the follow-up treatment of well-differentiated thyroid cancer. Tijdschrift voor nucleaire geneeskunde. 1999;21:98–100.
- Vallejo JA, Muros MA. Cost-effectiveness of using recombinant human thyroid-stimulating hormone before radioiodine ablation for thyroid cancer treatment in Spanish hospitals. Revista Española de Medicina Nuclear e Imagen Molecular (English Edition). 2017;36(6):362–370. doi: 10.1016/j.remnie.2017.09.001
- Luster M, Felbinger R, Dietlein M, Reiners C. Thyroid hormone withdrawal in patients with differentiated thyroid carcinoma: a one hundred thirty-patient pilot survey on consequences of hypothyroidism and a pharmacoeconomic comparison to recombinant thyrotropin administration. Thyroid. 2005;15(10):1147–1155. doi: 10.1089/thy.2005.15.1147
- Rosario PW, Xavier AC, Calsolari MR. Recombinant human thyrotropin in thyroid remnant ablation with 131I in high-risk patients. Thyroid. 2010;20(11):1247–1252. doi: 10.1089/thy.2010.0114
- Iizuka Y, Katagiri T, Ogura K, Inoue M, et al. Comparison of thyroid hormone withdrawal and recombinant human thyroid-stimulating hormone administration for adjuvant therapy in patients with intermediate- to high-risk differentiated thyroid cancer. Annals of Nuclear Medicine. 2020;34(10):736-741. doi: 10.1007/s12149-020-01497-0
- Robbins RJ, Driedger A, Magner J; The U.S. and Canadian Thyrogen Compassionate Use Program Investigator Group. Recombinant human thyrotropin-assisted radioiodine therapy for patients with metastatic thyroid cancer who could not elevate endogenous thyrotropin or be withdrawn from thyroxine. Thyroid. 2006;16(11):1121–1130. doi: 10.1089/thy.2006.16.1121
- Tu J, Wang S, Huo Z, et al. Recombinant human thyrotropin-aided versus thyroid hormone withdrawal-aided radioiodine treatment for differentiated thyroid cancer after total thyroidectomy: a meta-analysis. Radiotherapy and Oncology. 2014;110(1):25–30. doi: 10.1016/j.radonc.2013.12.018
- Ma C, Xie J, Liu W, et al. Recombinant human thyrotropin (rhTSH) aided radioiodine treatment for residual or metastatic differentiated thyroid cancer. Cochrane Database of Systematic Reviews. 2010. doi: 10.1002/14651858.CD008302
- Wolfson RM, Rachinsky I, Morrison D, et al. Recombinant Human Thyroid Stimulating Hormone versus Thyroid Hormone Withdrawal for Radioactive Iodine Treatment of Differentiated Thyroid Cancer with Nodal Metastatic Disease. Journal of Oncology. 2016:1–6. doi: 10.1155/2016/6496750
- Klubo-Gwiezdzinska J, Burman KD, Van Nostrand D, et al. Treatment of metastatic thyroid cancer: relative efficacy and side effect profile of preparation by thyroid hormone withdrawal versus recombinant human thyrotropin. Thyroid. 2012;22(3):310–317. doi: 10.1089/thy.2011.0235
- Wolffenbuttel BH, Coppes MH, Bongaerts AH, Glaudemans AW, Links TP. Unexpected symptoms after rhTSH administration due to occult thyroid carcinoma metastasis. The Netherlands journal of medicine. 2013;71(5):253–256.
- Tsai HC, Ho KC, Chen SH, et al. Feasibility of Recombinant Human TSH as a Preparation for Radioiodine Therapy in Patients with Distant Metastases from Papillary Thyroid Cancer: Comparison of Long-Term Survival Outcomes with Thyroid Hormone Withdrawal. Diagnostics. 2022;12(1):221 doi: 10.3390/diagnostics12010221
- Goldberg LD, Ditchek NT. Thyroid carcinoma with spinal cord compression. JAMA: The Journal of the American Medical Association. 1981;245(9):953-954. doi: 10.1001/jama.1981.03310340043025
- Hoelting T, Tezelman S, Siperstein AE, Duh QY, Clark OH. Biphasic effects of thyrotropin on invasion and growth of papillary and follicular thyroid cancer in vitro. Thyroid. 1995;5(1):35–40. doi: 10.1089/thy.1995.5.35
- Pietz L, Michałek K, Waśko R, et al. Wpływ stymulacji endogennego TSH na wzrost resztkowej objetości tarczycy u chorych po całkowitej tyreoidektomii z powodu raka zróznicowanego tarczycy. Endokrynologia Polska. 2008;59:119–122. (In Polish)
- Dedov II, Rumyantsev PO, Nizhegorodova KS, et al. Recombinant human thyrotropin in radioiodine diagnostics and radioiodine ablation of patients with well-differentiated thyroid cancer: the first experience in Russia. Endocrine Surgery. 2018;12(3):128–139. (In Russ). doi: 10.14341/serg9806
- Saracyn M, Lubas A, Bober B, et al. Recombinant human thyrotropin worsens renal cortical perfusion and renal function in patients after total thyroidectomy due to differentiated thyroid cancer. Thyroid. 2020;30(5):653–660. doi: 10.1089/thy.2019.0372
- Chaker L, Razvi S, Bensenor IM, et al. Hypothyroidism. Nature Reviews Disease Primers. 2022;8(1). doi: 10.1038/s41572-022-00357-7
- Ortiga-Carvalho TM, Sidhaye AR, Wondisford FE. Thyroid hormone receptors and resistance to thyroid hormone disorders. Nature Reviews Endocrinology. 2014;10(10):582–591. doi: 10.1038/nrendo.2014.143
- Lien EA, Nedrebo BG, Varhaug JE, et al. Plasma total homocysteine levels during short-term iatrogenic hypothyroidism. Journal of Clinical Endocrinology and Metabolism. 2000;85(3):1049–1053. doi: 10.1210/jcem.85.3.6439
- Bicikova M, Hampl R, Hill M, et al. Steroids, sex hormone-binding globulin, homocysteine, selected hormones and markers of lipid and carbohydrate metabolism in patients with severe hypothyroidism and their changes following thyroid hormone supplementation. Clinical Chemistry and Laboratory Medicine. 2003;41(3):284–292. doi: 10.1515/CCLM.2003.045
- Lee SJ, Lee HY, Lee WW, Kim SE. The effect of recombinant human thyroid stimulating hormone on sustaining liver and renal function in thyroid cancer patients during radioactive iodine therapy. Nuclear Medicine Communications. 2014;35(7):727–732. doi: 10.1097/MNM.0000000000000118
- Targher G, Montagnana M, Salvagno G, et al. Association between serum TSH, free T4 and serum liver enzyme activities in a large cohort of unselected outpatients. Clinical Endocrinology. 2008;68(3):481–484. doi: 10.1111/j.1365-2265.2007.03068.x
- Pearce EN, Wilson PW, Yang Q, Vasan RS, Braverman LE. Thyroid function and lipid subparticle sizes in patients with short-term hypothyroidism and a population-based cohort. The Journal of Clinical Endocrinology & Metabolism. 2008;93(3):888–894. doi: 10.1210/jc.2007-1987
- Ness GC, Lopez D, Chambers CM, et al. Effects of L-triiodothyronine and the thyromimetic L-94901 on serum lipoprotein levels and hepatic low-density lipoprotein receptor, 3-hydroxy-3- methylglutaryl coenzyme A reductase, and apo A-I gene expression. Biochemical Pharmacology. 1998;56(1):121–129. doi: 10.1016/S0006-2952(98)00119-1
- Pattaravimonporn N, Chaikijurajai T, Chamroonrat W, Sriphrapradang C. Myxedema Psychosis after Levothyroxine Withdrawal in Radioactive Iodine Treatment of Differentiated Thyroid Cancer: A Case Report. Case Reports in Oncology. 2021;14(3):1596–1600. doi: 10.1159/000520128
- Nagamachi S, Jinnouchi S, Nishii R, et al. Cerebral blood flow abnormalities induced by transient hypothyroidism after thyroidectomy – analysis by tc-99m-HMPAO and SPM96. Annals of Nuclear Medicine. 2004;18(6):469–477. doi: 10.1007/BF02984562
- Constant EL, De Volder AG, Ivanoiu A, et al. Cerebral blood flow and glucose metabolism in hypothyroidism: a positron emission tomography study. Journal of Clinical Endocrinology and Metabolism. 2001;86(8):3864–3870. doi: 10.1210/jcem.86.8.7749
- Duntas LH, Biondi B. Short-term hypothyroidism after Levothyroxine-withdrawal in patients with differentiated thyroid cancer: clinical and quality of life consequences. European Journal of Endocrinology. 2007;156(1):13–19. doi: 10.1530/eje.1.02310
- Kao PF, Lin JD, Chiu CT, et al. Gastric emptying function changes in patients with thyroid cancer after withdrawal of thyroid hormone therapy. Journal of Gastroenterology and Hepatology. 2004;19(6):655–660. doi: 10.1111/j.1440-1746.2003.03326.x
- Botella-Carretero JI, Prados A, Manzano L, et al. The effects of thyroid hormones on circulating markers of cell-mediated immune response, as studied in patients with differentiated thyroid carcinoma before and during thyroxine withdrawal. European Journal of Endocrinology. 2005;153(2):223–230. doi: 10.1530/eje.1.01951
- Duranton F, Lacoste A, Faurous P, et al. Exogenous thyrotropin improves renal function in euthyroid patients, while serum creatinine levels are increased in hypothyroidism. Clinical Kidney Journal. 2013;6(5):478–483. doi: 10.1093/ckj/sft092
- Coura-Filho GB, Willegaignon J, Buchpiguel CA, Sapienza MT. Effects of thyroid hormone withdrawal and recombinant human thyrotropin on glomerular filtration rate during radioiodine therapy for well-differentiated thyroid cancer. Thyroid. 2015;25(12):1291–1296. doi: 10.1089/thy.2015.0173
- An YS, Lee J, Kim HK, Lee SJ, Yoon JK. Effect of withdrawal of thyroid hormones versus administration of recombinant human thyroid-stimulating hormone on renal function in thyroid cancer patients. Scientific Reports. 2023;13(1). doi: 10.1038/s41598-023-27455-0
- Den Hollander JG, Wulkan RW, Mantel MJ, Berghout A. Correlation between severity of thyroid dysfunction and renal function. Clinical Endocrinology. 2005;62(4):423–427. doi: 10.1111/j.1365-2265.2005.02236.x
- Cho YY, Kim SK, Jung JH, et al. Long-term outcomes of renal function after radioactive iodine therapy for thyroid cancer according to preparation method: thyroid hormone withdrawal vs. recombinant human thyrotropin. Endocrine. 2019;64(2):293–298. doi: 10.1007/s12020-018-1807-x
- Kreisman SH, Hennessey JV. Consistent Reversible Elevations of Serum Creatinine Levels in Severe Hypothyroidism. Archives of Internal Medicine. 1999;159(1):79–82. doi: 10.1001/archinte.159.1.79
- Mariani LH, Berns JS. The Renal Manifestations of Thyroid Disease. Journal of the American Society of Nephrology. 2012;23(1):22–26. doi: 10.1681/ASN.2010070766
- Kim SK, Yun GY, Kim KH. et al. Severe hyponatremia following radioactive iodine therapy in patients with differentiated thyroid cancer. Thyroid. 2014;24(4):773–777. doi: 10.1089/thy.2013.0110
- Nozu T, Yoshida Y, Ohira M, Okumura T. Severe hyponatremia in association with I (131) therapy in a patient with metastatic thyroid cancer. Internal Medicine. 2011;50(19):2169–2174. doi: 10.2169/internalmedicine.50.5740
- Shakir MK, Krook LS, Schraml FV, Clyde PW. Symptomatic hyponatremia in association with a low-iodine diet and levothyroxine withdrawal prior to I131 in patients with metastatic thyroid carcinoma. Thyroid. 2008;18(7):787–792. doi: 10.1089/thy.2008.0050
- Al Nozha OM, Vautour L, How J. Life-threatening hyponatremia following a low-iodine diet: a case report and review of all reported cases. Endocrine Practice. 2011;17(5):e113–e117. doi: 10.4158/EP11045.CR
- Lee JE, Kim SK, Han KH, et al. Risk factors for developing hyponatremia in thyroid cancer patients undergoing radioactive iodine therapy. PLoS One. 2014;9(8):e106840. doi: 10.1371/journal.pone.0106840
- Horie I, Ando T, Imaizumi M, Usa T, Kawakami A. Hyperkalemia develops in some thyroidectomized patients undergoing thyroid hormone withdrawal in preparation for radioactive iodine ablation for thyroid carcinoma. Endocrine Practice. 2015;21(5):488–494. doi: 10.4158/EP14532.OR
- Hyer S, Kong A, Pratt B, Harmer C. Salivary gland toxicity after radioiodine therapy for thyroid cancer. Clinical Oncology. 2007;19(1):83–86. doi: 10.1016/j.clon.2006.11.005
- Riachy R, Ghazal N, Haidar MB, Elamine A, Nasrallah MP. Early Sialadenitis After Radioactive Iodine Therapy for Differentiated Thyroid Cancer: Prevalence and Predictors. International Journal of Endocrinology. 2020;2020:1–7. doi: 10.1155/2020/8649794
- Adramerinas M, Andreadis D, Vahtsevanos K, Poulopoulos A, Pazaitou-Panayiotou K. Sialadenitis as a complication of radioiodine therapy in patients with thyroid cancer: where do we stand? Hormones. 2021;20(4):669–678. doi: 10.1007/s42000-021-00304-3
- Silberstein E. Prevention of radiation sialadenitis and glossitis after radioiodine-131 therapy of thyroid cancer. Journal of Nuclear Medicine. 2007;48 Suppl. 2.
- Ma C, Xie J, Jiang Z, Wang G, Zuo S. Does amifostine have radioprotective effects on salivary glands in high-dose radioactive iodine-treated differentiated thyroid cancer. European Journal of Nuclear Medicine and Molecular Imaging. 2010;37(9):1778–1785. doi: 10.1007/s00259-009-1368-6
- Nakada K, Ishibashi T, Takei T. Does lemon candy decrease salivary gland damage after radioiodine therapy for thyroid cancer? Journal of nuclear medicine. 2005;46(2):261–266.
- Le Roux MK, Graillon N, Guyot L. Salivary side effects after radioiodine treatment for differentiated papillary thyroid carcinoma: Long-term study. Head & Neck. 2020;42(11):3133–3140. doi: 10.1002/hed.26359
- Jentzen W, Balschuweit D, Schmitz J, et al. The influence of saliva flow stimulation on the absorbed radiation dose to the salivary glands during radioiodine therapy of thyroid cancer using (124) I PET(/CT) imaging. European Journal of Nuclear Medicine and Molecular Imaging. 2010;37(12):2298–2306. doi: 10.1007/s00259-010-1532-z
- Trukhin AA, Yartsev VD, Sheremeta MS, et al. Nasolacrimal Duct Obstruction Secondary to Radioactive Iodine-131 Therapy for Differentiated Thyroid Cancer. The Bulletin of the Scientific Centre for Expert Evaluation of Medicinal Products. Regulatory Research and Medicine Evaluation. 2022;12(4):415–424. doi: 10.30895/1991-2919-2022-12-4-415-424
- Iakovou I, Goulis DG, Tsinaslanidou Z,et al. Effect of recombinant human thyroid-stimulating hormone or levothyroxine withdrawal on salivary gland dysfunction after radioactive iodine administration for thyroid remnant ablation. Head & Neck. 2016;38 Suppl. 1:E227–230. doi: 10.1002/hed.23974
- Rosario PW, Calsolari MR. Salivary and lacrimal gland dysfunction after remnant ablation with radioactive iodine in patients with differentiated thyroid carcinoma prepared with recombinant human thyrotropin. Thyroid. 2013;23(5):617–619. doi: 10.1089/thy.2012.0050
- Molenaar RJ, Sidana S, Radivoyevitch T, et al. Risk of Hematologic Malignancies After Radioiodine Treatment of Well-Differentiated Thyroid Cancer. Journal of Clinical Oncology. 2018;36(18):1831–1839. doi: 10.1200/JCO.2017.75.0232
- Signore A, Campagna G, Marinaccio J, et al. Analysis of Short-Term and Stable DNA Damage in Patients with Differentiated Thyroid Cancer Treated with 131I in Hypothyroidism or with Recombinant Human Thyroid-Stimulating Hormone for Remnant Ablation. Journal of Nuclear Medicine. 2022;63(10):1515–1522. doi: 10.2967/jnumed.121.263442
- Sohn SY, Choi JY, Jang HW, et al. Association between excessive urinary iodine excretion and failure of radioactive iodine thyroid ablation in patients with papillary thyroid cancer. Thyroid. 2013;23(6):741–747. doi: 10.1089/thy.2012.0136
- Lakshmanan M, Schaffer A, Robbins J, Reynolds J, Norton J. A simplified low iodine diet in I-131 scanning and therapy of thyroid cancer. Clinical Nuclear Medicine. 1988;13(12):866–868. doi: 10.1097/00003072-198812000-00003
- Maxon HR, Thomas SR, Boehringer A, et al. Low iodine diet in I-131 ablation of thyroid remnants. Clinical Nuclear Medicine. 1983;8(3):123–126. doi: 10.1097/00003072-198303000-00006
- Tala Jury HP, Castagna MG, Fioravanti C, et al. Lack of association between urinary iodine excretion and successful thyroid ablation in thyroid cancer patients. Journal of Clinical Endocrinology and Metabolism. 2010;95(1):230–237. doi: 10.1210/jc.2009-1624
- Lee M, Lee YK, Jeon TJ, et al. Low iodine diet for one week is sufficient for adequate preparation of high dose radioactive iodine ablation therapy of differentiated thyroid cancer patients in iodine-rich areas. Thyroid. 2014;24(8):1289–1296. doi: 10.1089/thy.2013.0695
- Tobey AE, Hongxiu L, Auh S, et al. Urine iodine excretion exceeding 250 ug/24h is associated with higher likelihood of progression in intermediate and high-risk thyroid cancer patients treated with radioactive iodine [abstract]. Thyroid. 2018;28 Suppl. 1:A40–A41
- Pluijmen MJ, Eustatia-Rutten C, Goslings BM, et al. Effects of low-iodide diet on postsurgical radioiodide ablation therapy in patients with differentiated thyroid carcinoma. Clinical Endocrinology. 2003;58(4):428–435. doi: 10.1046/j.1365-2265.2003.01735.x
- Morris LF, Wilder MS, Waxman AD, Braunstein GD. Reevaluation of the impact of a stringent low-iodine diet on ablation rates in radioiodine treatment of thyroid carcinoma. Thyroid. 2001;11(8):749–755. doi: 10.1089/10507250152484583
- Yoo IDKS, Kim SH, Seo YY, et al. The success rate of initial (131) i ablation in differentiated thyroid cancer: comparison between less strict and very strict low iodine diets. Nuclear Medicine and Molecular Imaging. 2012;46(1):34–40. doi: 10.1007/s13139-011-0111-y
- Ito S, Iwano S, Kato K, Naganawa S. Predictive factors for the outcomes of initial I-131 low-dose ablation therapy to Japanese patients with differentiated thyroid cancer. journal article. Annals of Nuclear Medicine. 2018;32(6):418–424. doi: 10.1007/s12149-018-1261-0
- Lim CY, Kim JY, Yoon MJ, et al. Effect of a low iodine diet vs. restricted iodine diet on postsurgical preparation for radioiodine ablation therapy in thyroid carcinoma patients. Yonsei Medical Journal. 2015;56(4):1021–1027. doi: 10.3349/ymj.2015.56.4.1021
- Kim HK, Lee SY, Lee JI, et al. Daily urine iodine excretion while consuming a low-iodine diet in preparation for radioactive iodine therapy in a high iodine intake area. Clinical Endocrinology. 2011;75(6):851–856. doi: 10.1111/j.1365-2265.2011.04157.x
- Tomoda C, Uruno T, Takamura Y, et al. Reevaluation of stringent low iodine diet in outpatient preparation for radioiodine examination and therapy. Endocrine Journal. 2005;52(2):237–240. doi: 10.1507/endocrj.52.237
- Sohaimi WF, Abdul Manap M, Kasilingam L, et al. Randomised controlled trial of one-week strict low-iodine diet versus one-week non-specified low iodine diet in differentiated thyroid carcinoma. Iranian Journal of Nuclear Medicine. 2019;27(2):99–105.
- Dekker BL, Links MH, Muller Kobold AC, et al. Low-Iodine Diet of 4 Days Is Sufficient Preparation for 131I Therapy in Differentiated Thyroid Cancer Patients. The Journal of Clinical Endocrinology & Metabolism. 2022;107(2):e604–e611. doi: 10.1210/clinem/dgab691
- Padovani RP, Maciel RM, Kasamatsu TS, et al. Assessment of the Effect of Two Distinct Restricted Iodine Diet Durations on Urinary Iodine Levels (Collected over 24 h or as a Single-Spot Urinary Sample) and Na (+)/I (-) Symporter Expression. European Thyroid Journal. 2015;4(2):99–105. doi: 10.1159/000433426
- Park JT, Hennessey JV. Two-week low iodine diet is necessary for adequate outpatient preparation for radioiodine rhTSH scanning in patients taking levothyroxine. Thyroid. 2004;14(1):57–63. doi: 10.1089/105072504322783858
- Dekker BL, Touw DJ, van der Horst-Schrivers ANA, et al. Use of Salivary Iodine Concentrations to Estimate the Iodine Status of Adults in Clinical Practice. The Journal of Nutrition. 2021;151(12):3671–3677. doi: 10.1093/jn/nxab303
Supplementary files